The inner fish speaks
« previous post | next post »
 One of the oldest and most interesting arguments for evolution is Ernst Haeckel's theory of recapitulation: the idea that ontogeny recapitulates phylogeny. In the form that Haeckel proposed — that embryological development progresses though a series of fully-developed ancestral forms — this theory has been refuted many times over the past century. What remains is the idea that "one species changes into another by a sequence of small modifications to its developmental program". This is the basis of modern research in evolutionary developmental biology ("evo devo"), and a central theme of Neil Shubin's Your Inner Fish, from which I took that picture of the development of arm bones from fish to humans.
One of the oldest and most interesting arguments for evolution is Ernst Haeckel's theory of recapitulation: the idea that ontogeny recapitulates phylogeny. In the form that Haeckel proposed — that embryological development progresses though a series of fully-developed ancestral forms — this theory has been refuted many times over the past century. What remains is the idea that "one species changes into another by a sequence of small modifications to its developmental program". This is the basis of modern research in evolutionary developmental biology ("evo devo"), and a central theme of Neil Shubin's Your Inner Fish, from which I took that picture of the development of arm bones from fish to humans.
Evo devo is mainly about anatomical development, but sometimes, surprising claims are made in this framework about evolutionary conservation of neurological function. A striking example of this is offered by a paper in the July 18 issue of Science (Andrew H. Bass, Edwin H. Gilland, and Robert Baker, "Evolutionary Origins for Social Vocalization in a Vertebrate Hindbrain–Spinal Compartment", Science 321(5887): 417-421, 2008), which argues that "the vocal basis for acoustic communication among vertebrates evolved from an ancestrally shared developmental compartment already present in the early fishes", namely "a segment-like region that forms a transitional compartment between the caudal hindbrain and rostral spinal cord".
This idea is a good deal harder to evoke with a picture than the evolution of arm bones is, though Bass et al. try. Here's their Fig. 1:
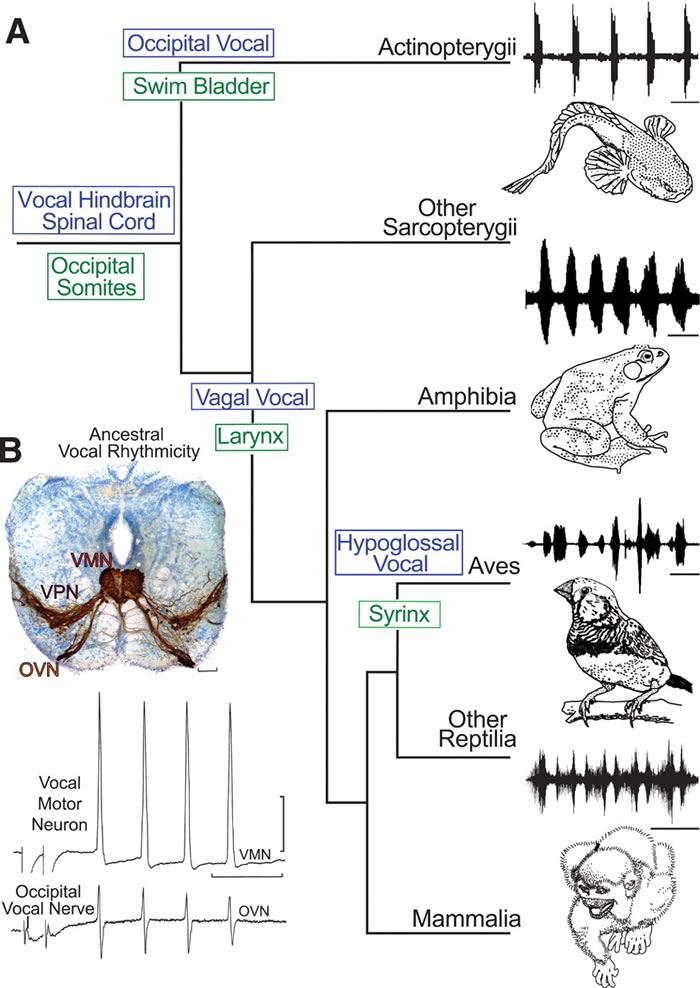
(A) Cladogram of living bony vertebrates (3), with oscillogram of a vocalization from a representative species, shows nodal (ancestral) states for vocal characters. Vocalizations (top to bottom): midshipman fish agonistic "grunts," bullfrog advertisement call, estrildid finch song, and squirrel monkey cackle. […] (B) Vocal pacemaker circuit. Among batrachoidid fish (midshipman and toadfish), there is a direct translation between the temporal properties of the vocal circuitry and natural calls. (Top) Transverse section at the caudal hindbrain-spinal cord transition of a toadfish shows transneuronal, neurobiotin-labeling of midline vocal motor neurons (VMNs), adjacent pacemaker neurons (VPN), and motor axons exiting via nerve root that gives rise to the occipital vocal nerve (OVN); vocal neurons have extensive lateral processes […] (Bottom) The rhythmic, oscillatory-like activity of a vocal motor neuron [(top trace) average of four DC-coupled intracellular records] is aligned with occipital nerve activity [(bottom trace) average of four intracranial records] to indicate relative timing.
The authors summarize the developmental pathways in Fig. 4:
(I) Schematic in horizontal plane showing relative positions of vocal and nonvocal neurons comprising a rhombomere (rh) 8 to spinal (SC) compartment. (J) Comparative summary in sagittal plane showing relative positions of vocal neurons in the rh8–spinal compartment. The developmental map for premotor-motor circuitry in fish (this report), as well as for motor neurons in birds and frogs, is based on representatives of similar age. Positioning of premotor neurons that participate in vocal patterning in birds and frogs, and motor and premotor neurons in mammals, including primates, is based on adult phenotypes … Abbreviations: Drt, dorsal reticular nucleus; PAm, nucleus parambigualis; Ri, inferior reticular formation; RAb, nucleus retroambiguus; RAm, nucleus retroambigualis; VPP-VPN-VMN, vocal prepacemaker–pacemaker–motor neurons; XMNc, caudal XMN; XIIts, tracheosyringeal division of hypoglossal motor nucleus.
The paper's main contribution was to trace the embryological development of vocal motor networks in Batrachoidid fish, such as toadfish and midshipman fish:
Although the genetic basis for human speech receives much attention, the fundamental issue of the ancestral origins of neural networks for vocal signaling is essentially unexplored. Social, context-dependent acoustic communication occurs in most of the major vertebrate lineages, including fishes. Teleost fish, the most species-rich of all vertebrate groups, have a simple repertoire of vocalizations complemented by vocal and auditory pathways that are organized similarly to those of amphibians, reptiles, birds, and mammals. Batrachoidid fish (midshipman and toadfish), in particular, have an expansive vocal-acoustic network, including a rhythmically firing, pacemaker–motor neuron circuit that directly determines the contraction rate of vocal muscles attached to the swim bladder and, in turn, the temporal properties of calls. Because batrachoidids also have readily studied larval stages, they were chosen to investigate the hypothesis that fish and terrestrial vertebrates share an ancestral origin of their vocal motor networks. Here, we show that the vocal systems of fishes and tetrapods develop very similarly in a segment-like region that forms a transitional compartment between the caudal hindbrain and rostral spinal cord.
The authors then argue for conservation of this basic developmental pathway for neural control of vocalization in frogs, reptiles, birds and mammals:
A taxonomic survey revealed a close alignment of the larval fish pattern with the vocal circuitry of other groups of vocal vertebrates. The position of the VMN compares well with that of developing vocal motor neurons that innervate the syrinx in birds and larynx in frogs. Although comparable developmental studies have not been done for reptiles and mammals, their adult phenotypes indicate a similar pattern for laryngeal motor neurons. Premotor neurons that pattern vocal-respiratory mechanisms in birds, amphibians, and mammals, including nonhuman primates and humans, are topographically comparable to the developing prepacemaker-pacemaker nuclei (VPP-VPN) of fish and, like the VPP-VPN, are targets of midbrain vocal neurons.
The most parsimonious interpretation of a cladistic analysis is that a vocal hindbrain-spinal compartment originated in a common ancestor of the two major groups of living fishes, the Actinopterygii, which includes the teleosts studied here, and the Sarcopterygii, which includes lobe-finned fishes (lungfish and coelacanth) and tetrapods.
This is somewhat surprising, given that the sound-generating organs and even the choice of nerves to connect to them are quite different across species, with quite different evolutionary origins:
The vocal muscles of fish and tetrapods also share origins from occipital somites, although the innervation of the vocal muscle associated with the sound-generating organ evolved at least three times: occipital nerve and swim bladder of fish (Actinopterygii); vagal nerve and larynx of nonavian tetrapods; and hypoglossal nerve and syrinx of birds.
But apparently some underlying circuitry for generating patterns in time is nevertheless conserved:
We conclude that our results reveal the ancestral origins of neural pattern generators for vocal-acoustic behaviors that mediate social signaling among all the major vertebrate lineages. […] Avian studies emphasize similarities with mammals in forebrain audio-vocal pathways. We propose that a more ancient neuroectodermal compartment, originating before the radiation of fishes that includes tetrapods, gave rise to the premotor-motor circuitry providing timing signals for a wide range of vertebrate acoustic behaviors.
Note that all of this involves very ancient and low-level parts of the brain, a lot older and deeper than the cerebral cortex. In primates, the relevant structures are the dorsal reticular nucleus, the nucleus retroambiguous, and the nucleus ambiguous. These are indeed involved in control of vocalization. Thus according to C.R. Larson, "Brain mechanisms involved in the control of vocalization", Journal of Voice 2:301-311:
The limbic system and diencephalon project to the midbrain periaqueductal gray (PAG) which may be important for coordination of various muscle groups involved in vocalization. The PAG neurons project to the reticular formation, nucleus retroambiguous, and nucleus ambiguous.
However, these same brainstem regions are also involved in breathing, swallowing, and some less obviously connected functions as well. Thus Peter O. Gerrits et al., "Retroambiguus Projections to the Cutaneus Trunci Motoneurons May Form a Pathway in the Central Control of Mating", J Neurophysiol 83: 3076-3083, 2000:
Our laboratory has proposed that the nucleus retroambiguus (NRA) generates the specific motor performance displayed by female cats during mating and that it uses direct pathways to the motoneurons of the lower limb muscles involved in this activity. In the hamster a similar NRA-projection system could generate the typical female mating posture, which is characterized by lordosis of the back as well as elevation of the tail. The present study attempted to determine whether this elevation of the tail is also part of the NRA-mating control system. […] The results indicate that during mating the NRA not only could generate the lordosis posture but also the elevation of the tail.
I'm not trying to suggest a deep connection between vocalization and mating postures — rather, my point is that in the brainstem just as in the cortex (and the genome), it's usually wrong to jump too quickly to the conclusion that there's a one-to-one correspondence between anatomical regions and humanly-salient functions.
But even if the nucleus retroambiguous is functionally, well, ambiguous, it's still interesting to learn that the developmental pathways of some of its functions may have been conserved through 440 million years of evolution, since the last common ancestor of the ray-finned and lobe-finned fishes.
[In support of their assertions about motor control of vocalizations in primates, Bass et al. cite Uwe Jürgens and Ludwig Ehrenreich, "The descending motorcortical pathway to the laryngeal motoneurons in the squirrel monkey", Brain Research 1148:90-95, 2007, which concludes that "the corticobulbar laryngeal control pathway synapses in the ipsilateral dorsal reticular nucleus and then divides into one component running directly to the ipsilateral nucleus ambiguus and a second component crossing to the contralateral nucleus ambiguus after having synapsed in the ipsilateral peri-ambigual reticular formation." Note however that overall motor control of the vocal organs (lips, tongue, velum etc.) involves a wide variety of pathways even at the brainstem level — for a review, see Uwe Jürgens, "Neural pathways underlying vocal control", Neuroscience and Biobehavioral Reviews, 26(2): 235-258, 2002, which provides this handy chart.
One other important difference, with respect to the mammalian pathway for laryngeal control that is apparently homologous to the toadfish system for swim-bladder sound generation: the toadfish hum is generated by rapid (up to 200 Hz) periodic contraction of a swim-bladder muscle, as coincidentally described by Natalie Angier a few days ago in the NYT ("Learning From a Muddy Muscle Master", 7/8/2008). I believe that the oscillogram in the bottom left of Fig. 1 (above) is showing the neural substrate for this rapid muscularly-generated hum. In contrast, mammals generate periodic ("voiced") sounds by passive oscillation of their vocal folds, using roughly the same physical mechanism as a vibrating oboe reed (or a burp), as described here. Basically, the combination of air pressure, tissue elasticity and Bernoulli forces creates a more-or-less stable oscillation. ]
Rob Gunningham said,
July 18, 2008 @ 9:27 am
I remember in Venice once being given Toadfish tail for dinner. It wasn't that special, much less remarkable than a talking fish.
Mark Liberman said,
July 18, 2008 @ 10:12 am
Rob Gunningham: I remember in Venice once being given Toadfish tail for dinner. It wasn't that special, much less remarkable than a talking fish.
The "supporting online material" for the Bass et al. paper provides three movies, described as follows:
As instances of "talking", these are not especially remarkable either. So maybe your toadfish-tail dinner was not such a bad bargain.
The Science article doesn't explain the difference between "type I" and "type II" males, but this 2005 article from the Berkeley Planet does:
Rob Gunningham said,
July 18, 2008 @ 11:07 am
As instances of "talking", these are not especially remarkable either. So maybe your toadfish-tail dinner was not such a bad bargain.
Nah. I still like the talking fish.
John Cowan said,
July 18, 2008 @ 12:59 pm
The phrase "nucleus ambiguous" is interesting because of its NA order. I would have expected "ambiguous nucleus" or a fully Latin "nucleus ambiguus" (or "ambiguosus", but L&S lists no such word).
Simon Fodden said,
July 18, 2008 @ 3:30 pm
Poor toadfish. Many years ago I had the unenviable task of catching and pithing toadfish (Opsanus tau) at Solomon's Island, Maryland, for my dad, who was doing research on diabetes. It seems that the beast has islets of Langerhans have that tissue separated as a discrete mass, and this was important for his work.
You couldn't have persuaded me to eat one — or even a portion of one — for love nor money, though. Now the soft shell crabs from the Bay… that's another thing.
Rob Gunningham said,
July 19, 2008 @ 7:40 am
Simon Fodden: Poor toadfish…it seems that the beast has islets of Langerhans have that tissue separated as a discrete mass, and this was important for his work.
And probably they were the only fish who were willing to talk about it.
Actually, this is interesting. Good for your dad!
Wednesday Round Up #22 « Neuroanthropology said,
July 29, 2008 @ 10:40 pm
[…] Log, The Inner Fish Speaks Vocal basis for speech in fish, built from evolutionary developmental biology? For the general […]